Richard Harris appears in the following:
Vitamin C Fails Again As Treatment For Sepsis
Wednesday, February 24, 2021
Sepsis is one of the most common causes of death for hospitalized patients. The search for an effective treatment has been frustrating.
Vitamin C Apparently Not Useful For Sepsis After All
Tuesday, February 23, 2021
A seemingly easy and cheap treatment for a deadly disease has failed in a major study. Vitamin C is apparently not useful for sepsis after all. That's a lesson for similar COIVD-19 treatments.
Long-Term Studies Of COVID-19 Vaccines Hurt By Placebo Recipients Getting Immunized
Friday, February 19, 2021
Researchers are trying to learn more about COVID-19 vaccines from original study participants. The quest is hampered because many people who first received a placebo shot are opting for the vaccine.
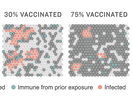
How Herd Immunity Works — And What Stands In Its Way
Thursday, February 18, 2021
Scientists say the pandemic will only end in the U.S. when we achieve what's called herd immunity. Play with our simulations to see how immunity can stop an outbreak in its tracks.
Long-Term COVID-19 Vaccine Studies Hampered As Placebo Recipients Get Real Shot
Wednesday, February 17, 2021
Scientists are trying to learn more about COVID-19 vaccines from original study participants. That quest is hampered because many people who received a placebo shot have now opted to get the vaccine.
COVID-19 Vaccines Could Add Fuel To Evolution Of Coronavirus Mutations
Wednesday, February 10, 2021
COVID-19 vaccines appear to work against the new coronavirus strains, though scientists are warily watching a variant first seen in South Africa. Vaccines may need updates to keep pace with the virus.
Vaccines Could Drive The Evolution Of More COVID-19 Mutants
Tuesday, February 09, 2021
Mutant coronaviruses can make vaccines less effective. At the same time, vaccines can contribute to virus mutations, but this is a slow process that should be manageable.
A Rocky Road On The Way To Herd Immunity For COVID-19
Wednesday, February 03, 2021
At least 70% of people will need to be immune from the coronavirus before COVID-19 can recede through a process known as herd immunity. Vaccines can play a role. But reaching the goal won't be easy.
After A Year Battling COVID-19, Drug Treatments Get A Mixed Report Card
Friday, January 22, 2021
Drugs for COVID-19 are sorted into three basic categories: They work, they don't work, or there simply isn't enough information to know. A generic steroid is one medicine that proved helpful.
3 Questions And The Emerging Answers About COVID-19 Vaccine Protection
Monday, January 18, 2021
First, can someone who has been vaccinated still spread the disease? Second, will the vaccine remain effective as the virus itself evolves? And third, how long will the vaccine's protection last?
Immune System Studies Help Answer Questions About COVID-19 Vaccine
Friday, January 15, 2021
There are many questions remaining about COVID-19 vaccines, such as how long protection will last and whether vaccinated people can spread the virus. Immunologists are working hard to get the answers.
Tracking Down Antibody Treatment Is A Challenge For COVID-19 Patients
Monday, January 11, 2021
Antibody-based drugs that bind to the coronavirus to prevent it from invading cells can help patients with mild to moderate COVID-19. But the medicines can be tough to find in time.
Doctors Encouraged By Antibody Treatments For COVID-19
Wednesday, January 06, 2021
While logistical challenges have hampered use of antibody drugs to treat people with mild-to-moderate COVID-19, recent results show the medicines can be worthwhile.
Doses Of Antibody Drugs Remain Unused As They Present Various Challenges
Tuesday, January 05, 2021
Monoclonal antibody drugs for COVID-19 with mild to moderate symptoms are not widely used, in part because doctors aren't sure they work. New data could provide more confidence in these drugs.
Why The COVID-19 Vaccine Distribution Has Gotten Off To A Slow Start
Friday, January 01, 2021
The rollout of vaccines for COVID-19 has been slower than expected. Reasons include local logistics, lack of funding and staffing struggles during the winter holidays.
1st Known U.S. Case Of U.K. Coronavirus Variant Found In Colorado
Wednesday, December 30, 2020
Colorado health officials found the variant of COVID-19 that spreads faster than the common strain. U.K. scientists identified the strain last week. It has spread in the U.K. and to other countries.
Low Demand For Antibody Drugs Against COVID-19
Tuesday, December 22, 2020
Monoclonal antibodies to prevent severe COVID-19 aren't being used as widely as expected. Medical staff shortages and patient transportation problems are two of the reasons.
Latest On FDA Authorization For Moderna's COVID-19 Vaccine
Friday, December 18, 2020
The Food and Drug Administration says it will soon grant emergency authorization to Moderna's COVID-19 vaccine. NPR shares the latest news.
Oxygen-Detecting Devices Give Misleading Readings In People With Dark Skin
Wednesday, December 16, 2020
Common devices to measure oxygen in the blood don't work as well in people with darker skin, according to a new study. They are useful, but experts warn readings should be interpreted more carefully.
Devices Used In COVID-19 Treatment Can Give Errors For Patients With Dark Skin
Wednesday, December 16, 2020
Fingertip devices that measure oxygen in the blood can sometimes give erroneous readings in people with dark skin, doctors report. The devices can say oxygen levels are normal when they're not.